Because ionic interactions involve the most charge they are the strongest intermolecular interactions that occur. Which of the following are the strongest molecular interactions.
Disulfide bridges hydrogen bonds Van der Waals interactions ionic bonds These bonds are all equally strong Question 7 2 points An alpha helix is a secondary structure of protein that is stabilized by peptide bonding between.

. Permanent dipole force d. Which molecule has the strongest intermolecular forces. Molecule Molecular Weight amu Dipole Boiling Moment Point Melting Point OH 600950 165 D 3555 K 185 K 881051 173 D 3502 K 190 K o F FIF S FIF F 146055 000 D 2223 K 2093 K F F -F 1200214 156 D 2247 K 1702 K F F 1061650 064 D 4175 K 249.
Which of the following is the strongest intermolecular interaction. Which of the following is the strongest bond. The interactions among the molecules in molecular solids are generally stronger than those among the particles that define either covalent or ionic lattices.
Is Van der Walls interactions Van der Walls interactions in a nonpolar environment covalent bonds ionic bonds in an aqueous environment or hydrogen bonds the strongest molecular interaction. Which type of bond is formed by atoms sharing electrons. Hydrophobic groups minimise their contact with water.
Label each statement as either a covalent or noncovalent interaction. B-hydrophobic groups interact through dispersion forces. London dispersion forces D.
All three have van der Waals induced dipole-induced dipole interactions between molecules due to the hydrocarbon groups. Which of the following interactions are the strongest. Pages 7 This preview shows page 1 - 3 out of 7 pages.
Which of the following interactions is the strongest. Which of the following is the strongest molecular force that causes a protein to. But the biodiesel molecule also has the electronegative oxygen atoms that make the molecule polar so that there are also dipole-dipole interactions between the molecules which are stronger than the van der Waals forces.
Hydrogen bonding force b. The strength of a hydrogen bond depends upon the electronegativities and sizes of the two atoms. Which of the following substances has the strongest intermolecular interactions.
Dipole-dipole interactions are the strongest intermolecular force of attraction. What is the strongest intermolecular force that occurs between methane CH4 and ammonia NH3. Hydrogen bonding force b.
The answer is hydrogen-bonding the which occurs when hydrogen is bound to a strongly electronegative element such as oxygen or nitrogen or. Chemmical reaction are molecular interactions so everything that is not made up of single atoms. A hydrogen atom between two small electronegative atoms such as F O N causes a strong intermolecular interaction known as the hydrogen bond.
Course Title CHEM 214. Which of the following compounds is expected to have the strongest interaction between its molecules. What is the strongest IMF for HI.
London dispersion forces b. Permanent dipole force d. An ion-dipole and a charge-induced dipole both involve a a a.
The strongest interactions are between ions Ionic interactions are attractive interactions that occur between oppositely charged ions that is atoms that carry a charge that is at least equal to the full charge of a proton or electron. Question 6 2 points Which of the following molecular interactions involved in protein tertiary structure is the strongest. CO2 NO2 SO2 H2S.
Ethanol C 2 H 5 OH and methyl ether CH 3 OCH 3 have the same molar mass. A - water molecules strongly interact with each other. Which of the following is the strongest molecular.

Hydrogen Bonding Strongest Fon Chemie Unbelebte Natur Lebewesen
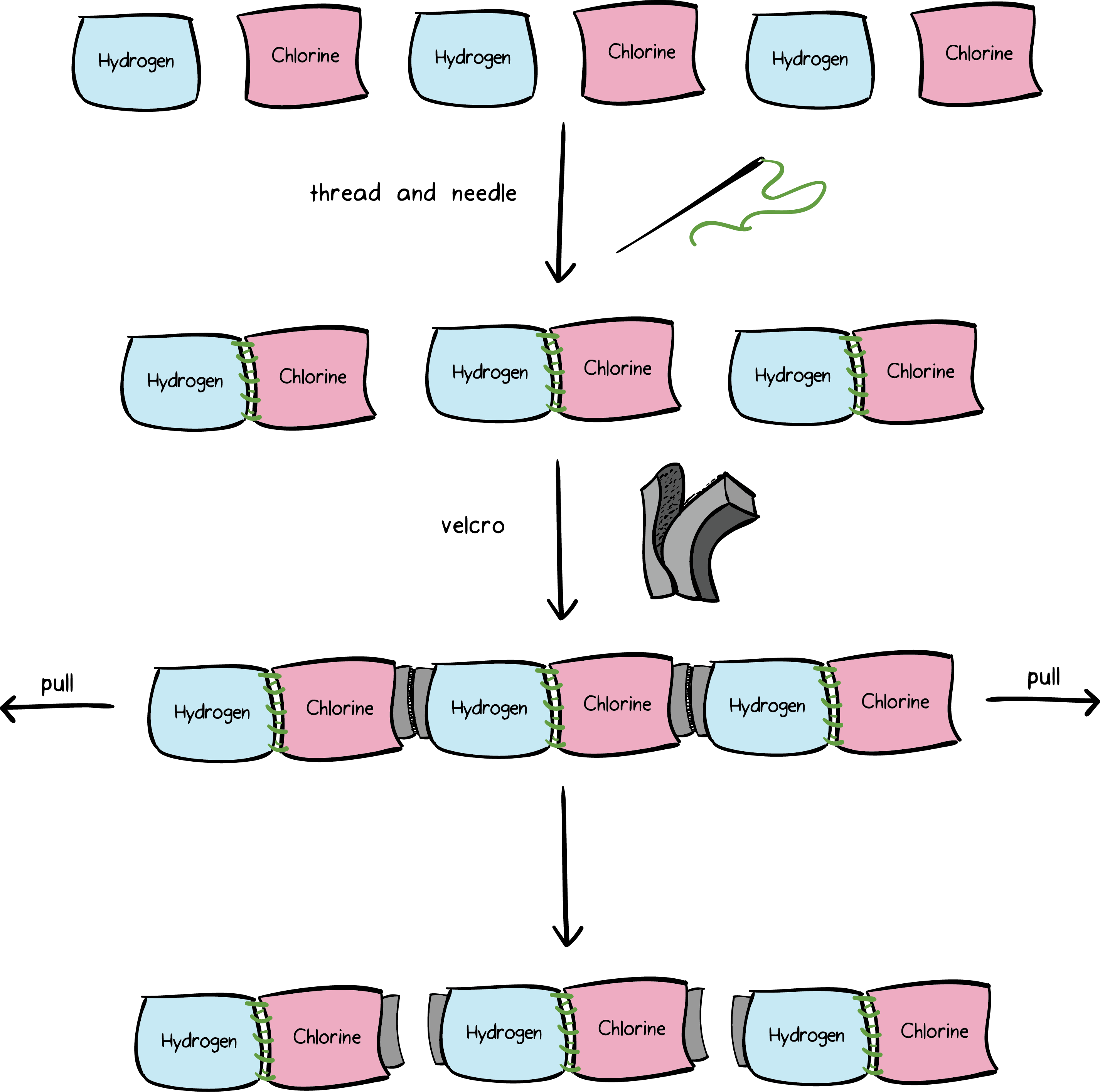
Intramolecular And Intermolecular Forces Article Khan Academy
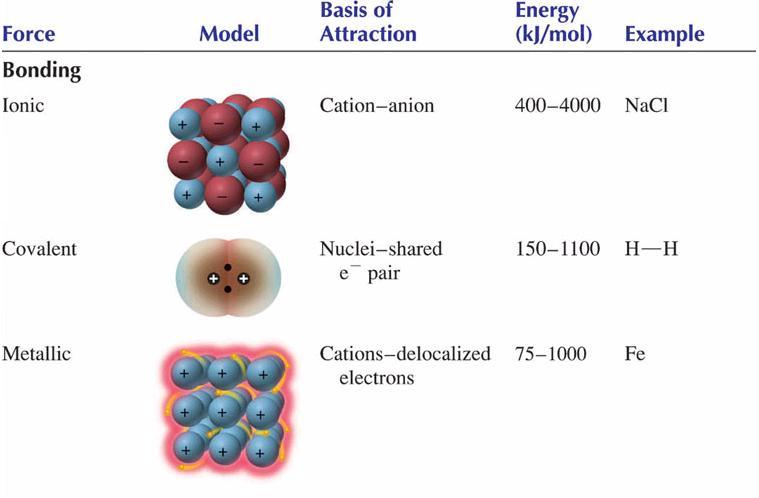
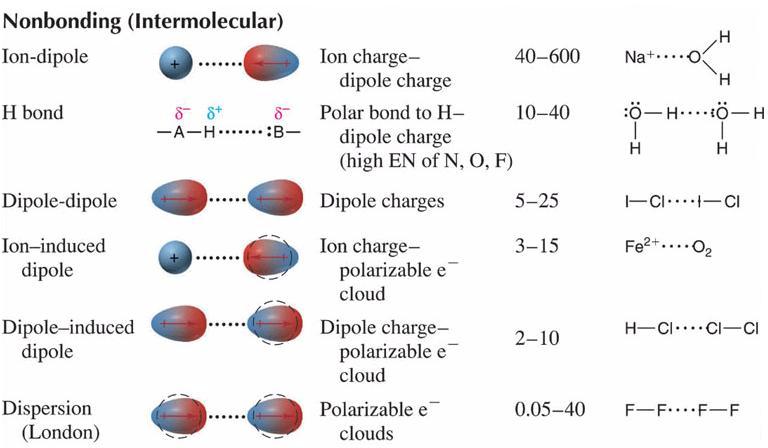
0 Comments